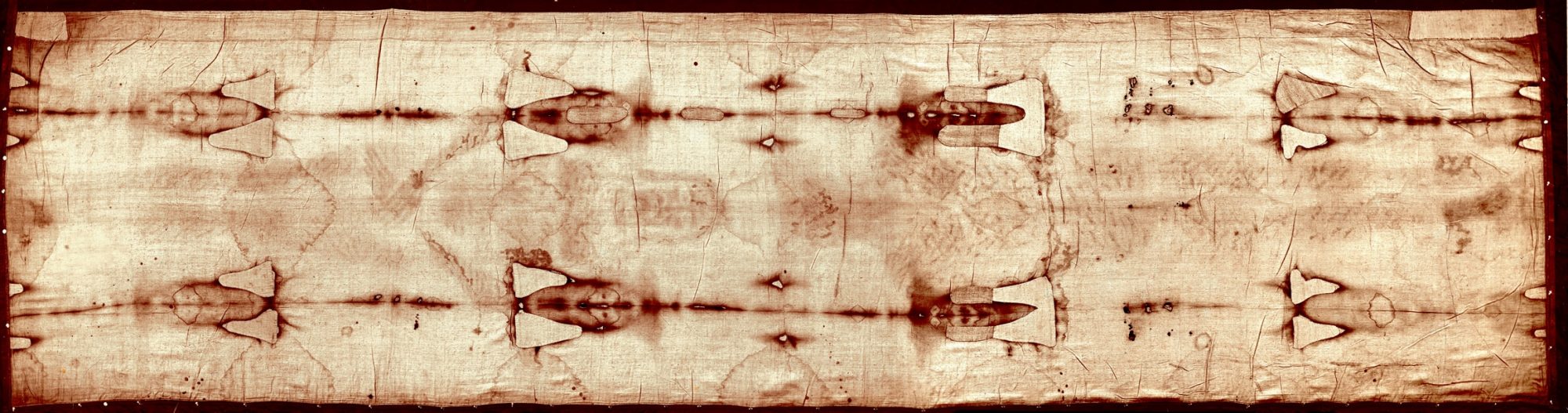
Ray Rogers theorized the image is the result of a Maillard reaction.
Raymond N. Rogers and Anna Arnoldi, in a joint paper of 2003 proposed that amines from a recently deceased human body may have undergone Maillard reactions with this carbohydrate layer within a reasonable period of time, before liquid decomposition products stained or damaged the cloth. The gases produced by a dead body are extremely reactive chemically, and within a few hours (in an environment such as a tomb) a body starts to produce heavier amines, such as putrescine and cadaverine, in its tissues.
https://en.wikipedia.org/wiki/Raymond_Rogers
His proposal would be a naturalistic explanation that is not artistic and involve a dead body.
We can now formally propose a completely natural hypothesis for image formation. Impurities in ancient linen could have been suspended by the surfactant property of a Saponaria officinalis washing solution and they would be concentrated at the cloth surface by evaporation. Reducing saccharides would react rapidly with the amine decomposition products of a dead body.
https://www.shroud.com/pdfs/rogers7.pdf
We see the result of the Maillard reaction in the browning of food.
The Maillard reaction is responsible for many colors and flavors in foods, such as the browning of various meats when seared or grilled, the browning and umami taste in fried onions and coffee roasting. It contributes to the darkened crust of baked goods, the golden-brown color of French fries and other crisps, browning of malted barley as found in malt whiskey and beer, and the color and taste of dried and condensed milk, dulce de leche, toffee, black garlic, chocolate, toasted marshmallows, and roasted peanuts
https://en.wikipedia.org/wiki/Maillard_reaction
Tanning of bodies in peat bogs is also from a Maillard reaction.
In archaeology the Maillard process occurs when bodies are preserved in peat bogs. The acidic peat environment causes a tanning or browning of skin tones and can turn hair to a red or ginger tone. The chemical mechanism is the same as in the browning of food, but it develops slowly over time due to the acidic action on the bog body. It is typically seen on Iron Age bodies and was described by Painter in 1991 as the interaction of anaerobic, acidic, and cold (typically 4 °C (39 °F)) sphagnum acid on the polysaccharides.
https://en.wikipedia.org/wiki/Maillard_reaction
Maillard reaction is a chemical reaction involving amino acids and sugars.
The Maillard reaction (/maɪˈjɑːr/ my-YAR; French: [majaʁ]) is a chemical reaction between amino acids and reducing sugars that gives browned food its distinctive flavor. Seared steaks, fried dumplings, cookies and other kinds of biscuits, breads, toasted marshmallows, and many other foods undergo this reaction.Caramelization is an entirely different process from Maillard browning, though the results of the two processes are sometimes similar to the naked eye (and taste buds). Caramelization may sometimes cause browning in the same foods in which the Maillard reaction occurs, but the two processes are distinct. They are both promoted by heating, but the Maillard reaction involves amino acids, whereas caramelization is the pyrolysis of certain sugars.[12]
https://en.wikipedia.org/wiki/Maillard_reaction
One feature the Maillard reaction explains is it can color the outside of the fibers without coloring the inside of fibers.
The coating of brown products is too thin to be resolved with a light microscope, and it is all on the outside of the fibres. There is no coloration in the medullas: the colour does not scorch the cellulose. There is essentially no colour on fibres from the middle of the back surface.
https://www.shroud.com/pdfs/rogers7.pdf
Though it can explain the coloring on fibers, it fails to explain many other features.
The TS image has high resolution. How can vapor from a body produce detailed imaging?
It also does not explain the head gap, imaging of the hair, depth encoding, angle encoding, x-ray effect, uniform density of ventral and dorsal sides, faint image on back of cloth, second degree distortions, unbroken blood stains, and uncolored fibers adjacent to colored fibers.
The fatal blow to the Maillard reaction theory is there is no sign of any decomposition of the body on the shroud. This evidence is contradictory to the Maillard reaction theory since ammonia gas is required for the Maillard reaction.
https://debatingchristianity.com/forum/viewtopic.php?p=1124081#p1124081