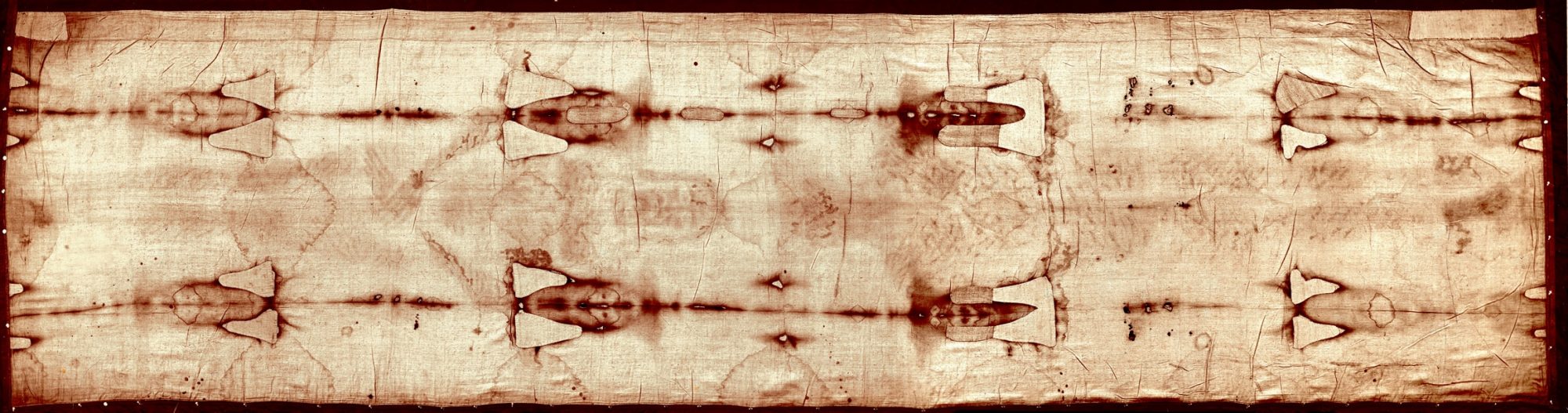
Calcium particles on the feet area of the shroud chemically match those in areas of Jerusalem.
In addition, a fiber taken from the foot-area of the Shroud’s body-image was tested for
calcium and was found to match the chemical composition of limestone found in
Jerusalem, in particular the limestone found at the burial sites of the Church of the Holy
Sepulcher and the Garden Tomb.
https://www.shroud.com/pdfs/sjoseph.pdf
The limestone found on the feet contains calcium in the form of aragonite. Similar
characteristics were found on samples coming from Ecole Biblique tomb in Jerusalem.
https://www.researchgate.net/publicatio … RIN_SHROUD
According to Kohlbeck the Jerusalem limestone … was primarily
travertine aragonite deposited from springs, rather than the more
common calcite. Calcite and aragonite differ in their crystalline structure – calcite
being rhombohedral and aragonite orthorhombic. Aragonite is less common than
calcite. Aragonite is formed under a much narrower range of conditions than calcite.
In addition to the aragonite, our Jerusalem samples also contained small quantities of
iron and strontium but no lead.We then examined a calcium sample from the Shroud taken from the area known as the
“bloody foot” because his showed a larger concentration of calcium carbonate than other
areas. This calcium carbonate turned out to be aragonite, not the more common calcite – and
exhibited small amounts of strontium and iron.Further analysis was conducted by Dr. Ricardo Levi-Setti, of the Enrico Fermi Institute of the
University of Chicago, who put both Shroud and Jerusalem samples through his high-
resolution scanning ion microprobe and produced graphs; these graphs revealed that the
samples were an unusually close match, except for minute pieces of flax that could not be
separated from the Shroud’s calcium and caused a slight organic variation.
https://www.shroud.com/pdfs/n14part3.pdf
If the TS was the result of a Medieval forgery, why would a forger go through the effort of getting dirt from a Jersusalem tomb? Especially since nobody would’ve discovered this level of detail until hundreds of years later.
https://debatingchristianity.com/forum/viewtopic.php?p=1120231#p1120231